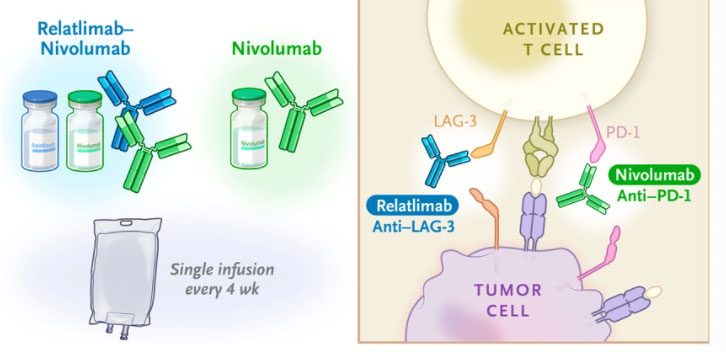
An immunotherapy study suggests hopeful results for advanced melanoma patients. The study indicated that relatlimab and nivolumab elicited beneficial responses in patients with late-stage melanoma. Specifically, the phase 2-3 trial indicated that combination therapy extended progression free-survival despite an individual’s prognostic indicators. Through using both antibodies to inhibit two immune checkpoint pathways, greater immune response was stimulated against cancer cells. Continued research is centered on the effects of combination therapy for patients in different stages of melanoma progression.
Novel drug combination may boost chemoimmunotherapy response in bladder cancer patients
A proof-of-concept study suggests that the combination of anti-inflammatory medication and chemotherapy drugs can boost immune response to suppress bladder tumor growth. Led by Cedars-Sinai Cancer investigators, the study focused on tackling the immune-dampening effect of chemotherapy drugs. Research uncovered the underlying mechanism that results in chemotherapy treatment failure, the release of prostaglandin E2. This bioactive lipid is associated with inflammation and inhibits dendritic maturation. A preceding factor for cells to fight cancer. In addition, early investigation revealed potential in combining celecoxib, an anti-inflammatory medication, and chemotherapy drugs as improved immune response was observed in treated mice models. Researchers look to test the efficacy of the potential treatment in human trials for bladder cancer patients.
Two-pronged immunotherapy treatment tested for effectiveness against glioblastomas
Recent clinical trials at the University of Cincinnati are examining the effectiveness of a two-pronged immunotherapy procedure to treat glioblastomas, aggressive brain tumors. Current treatment involves the combination of surgery, radiation, and chemotherapy, as to ensure microscopic particles of cancerous tissue do not survive. But despite continued advancements over the past decade, patients with glioblastomas remain uncured. Current clinical trials utilize a two-pronged approach, administering a combination of two immunotherapy drugs in tandem, that target different immune checkpoint proteins to activate immune cells. Previous single immunotherapy treatment observed failure led to the two-pronged approach in current trials.