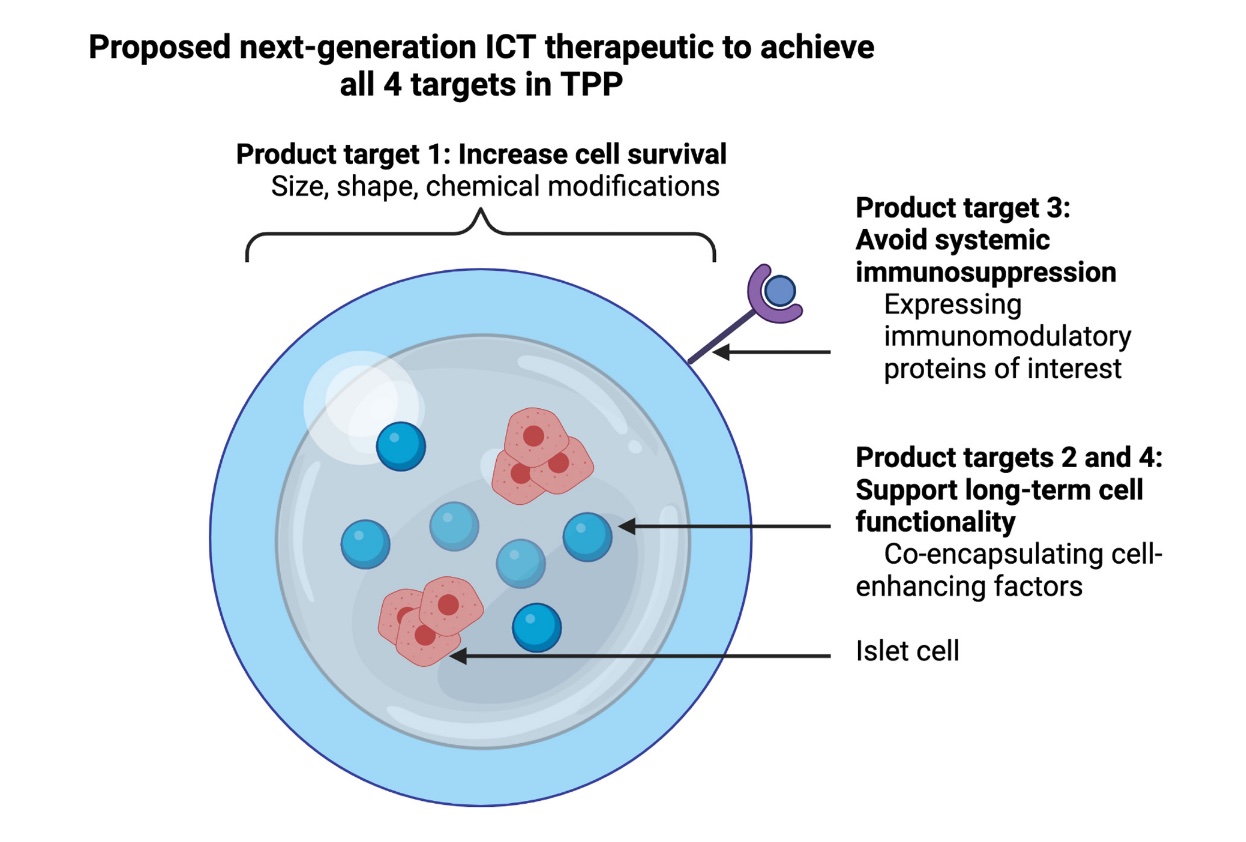
Despite optimal insulin replacement therapy, more than 40% of patients living with type 1 diabetes (T1D) experience severe hypoglycemic reactions and many others experience complications of their disease like vascular and renal damage. These patients require less burdensome and potentially curative approaches for their T1D, most notably including islet cell transplantation (ICT). Previous clinical testing proved the novel therapy successful in initial establishment of insulin independence but low islet viability, functionality, and localized immune protection limits ICT currently as a therapeutic for T1D. In order to address these limitations, Dr. Fatma Dogan and her team at VIC present a review on potential solutions that challenge ICT and promising combinatorial approaches that can optimize this therapeutic approach while clearly targeting the desired clinical outcomes.
Recent Publication in the Journal of Inflammation from Dr. Ruxandra Sîrbulescu
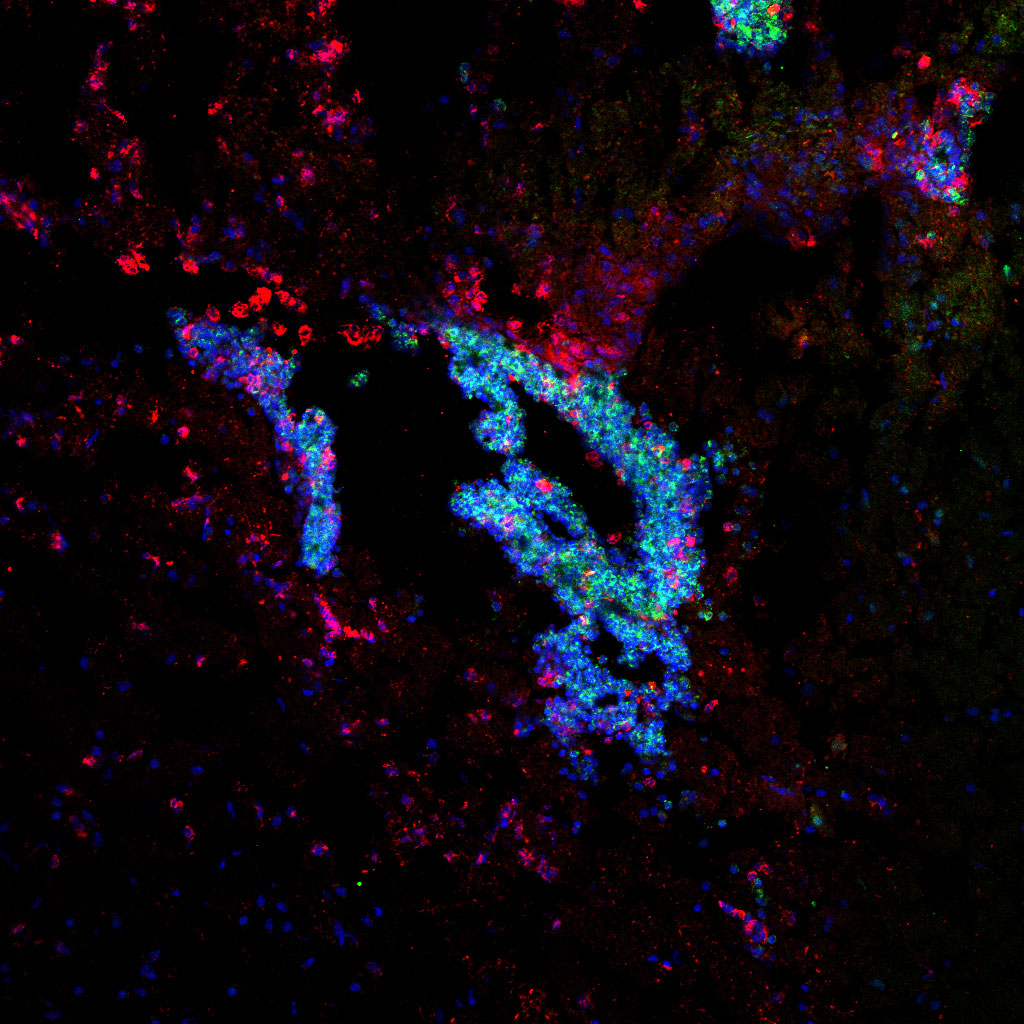
In a recent publication, Dr. Ruxandra Sîrbulescu and her team at VIC investigate the mechanisms that underlie the neuroprotective effects of a novel cell-based treatment that uses B cells to reduce the detrimental impact of inflammation after brain injury. The authors map the local cellular responses in the injured tissue along a 2-month timeline, and describe some of the key ways by which the applied therapeutic B cells communicate with and regulate the responses of inflammatory cells in their vicinity. The study indicates that B cells applied directly into the injured tissue regulate the actions of other immune cells, including monocytes and macrophages that infiltrate from the blood, resulting in less damage to the nervous tissue. The presence of therapeutically administered B cells also reduced the long-term activation of microglia, resident immune cells of the brain. Together, these findings support B cells as powerful regulatory cells with potential for further development as an effective cell therapy for treating traumatic brain injuries in the future.
Congratulations to Dr. Ruxandra F. Sîrbulescu for her Promotion to Assistant Professor at Harvard Medical School
Congratulations to Dr. Ruxandra F. Sîrbulescu, Principal Investigator at VIC, for her promotion to Assistant Professor in Neurology at Harvard Medical School! Dr. Sîrbulescu and her team at VIC focus on understanding mechanisms of neuroinflammation and cell-based immunomodulation in the context of acute and chronic brain injury and neurodegenerative diseases. In a recent article published in the Journal of Neuroinflammation, Dr. Sîrbulescu and her team investigate mechanisms of immunomodulation in the context of traumatic brain injury using local therapeutic application of B cells, and demonstrate that in the appropriate setting, these immune cells can act as powerful neuroprotective agents. Dr. Sîrbulescu’s innovative research has been recognized through multiple publications, media articles, patents, and distinctions, including the Dr. Melvin Field Innovation Award, and several Excellence in Innovation certificates from Partners Healthcare, as well as funding support from the National Institutes of Health.